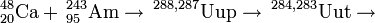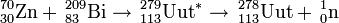Ununtrium
2008/9 Schools Wikipedia Selection. Related subjects: Chemical elements
|
|||||||||||||||||||||||||||||||
| General | |||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Name, Symbol, Number | ununtrium, Uut, 113 | ||||||||||||||||||||||||||||||
| Chemical series | presumably poor metals | ||||||||||||||||||||||||||||||
| Group, Period, Block | 13, 7, p | ||||||||||||||||||||||||||||||
| Appearance | unknown, probably silvery white or metallic gray |
||||||||||||||||||||||||||||||
| Standard atomic weight | [284] g·mol−1 | ||||||||||||||||||||||||||||||
| Electron configuration | perhaps [Rn] 5f14 6d10 7s2 7p1 (guess based on thallium) |
||||||||||||||||||||||||||||||
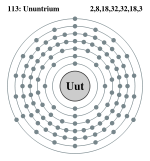
| Electrons per shell | 2, 8, 18, 32, 32, 18, 3 | ||||||||||||||||||||||||||||||
| Phase | presumably a solid | ||||||||||||||||||||||||||||||
| CAS registry number | 54084-70-7 | ||||||||||||||||||||||||||||||
| Selected isotopes | |||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||
Ununtrium (pronounced /juːˈnʌntriəm/ or /əˈnʌntriəm/) is the temporary name of a synthetic element in the periodic table that has the temporary symbol Uut and has the atomic number 113.
It has been synthesised both directly in "cold" and "warm" fusion reactions. It was first observed in the decay of ununpentium. Only eight atoms of ununtrium have been observed to date. Following periodic trends it is expected to be a soft, silvery metal.
Discovery Profile
The first report of ununtrium was in August 2003 when it was identified as a decay product of ununpentium. These results were published on February 1, 2004, by a team composed of Russian scientists at Dubna ( Joint Institute for Nuclear Research), and American scientists at the Lawrence Livermore National Laboratory.
On July 23, 2004, a team of Japanese scientists at RIKEN detected a single atom of 278Uut using the cold fusion reaction between Bismuth-209 and zinc-70. They published their results on September 28, 2004.
Support for their claim appeared in 2004 when scientists at the Institute of Modern Physics (IMP) identified 266Bh as decaying with identical properties to their single event (see bohrium).
The RIKEN team produced a further atom on April 2, 2005, although the decay data was different from the first chain, and may be due to the formation of a meta-stable isomer.
The Dubna-Livermore collaboration has strengthened their claim for the discovery of ununtrium by conducting chemical experiments on the decay daughter 268Db. In experiments in Jun 2004 and Dec 2005, the Dubnium isotope was successfully identified by milking the Db fraction and measuring any SF activities. Both the half-life and decay mode were confirmed for the proposed 268Db which lends support to the assignment of Z=115 and Z=113 to the parent and daughter nuclei.
Theoretical estimates of alpha-decay half-lives of alpha-decay chains from element 113 are in good agreement with the experimental data.
Naming
Current names
The element with Z=113 is historically known as eka-thallium. Ununtrium (Uut) is a temporary IUPAC systematic element name. Research scientists usually refer to the element simply as element 113 (E113).
Proposed names by claimants
Claims to the discovery of element 113 have been put forward by Dmitriev of the Dubna team and Morita of the RIKEN team. The JWP will decide to whom the right to suggest a name will be given. The IUPAC have the final say on the official adoption of a name. The table below gives the names that the teams above have suggested and which can be verified by press interviews.
| Group | Proposed Name | Proposed Symbol | Derivation |
|---|---|---|---|
| RIKEN | Japonium | Jp | Japan - country of group claimants |
| RIKEN | Rikenium | Rk | RIKEN - institute of group claimants |
Disallowed names
According to IUPAC rules, names used for previous elements that have ultimately not been adopted are not allowed to be proposed for future use. The table below summarises those names which are probably not allowed to be proposed by the claimant laboratories under the rules.
| Group Claim Affected | Disallowed Name | Disallowed Symbol | Reason |
|---|---|---|---|
| RIKEN | Nipponium | Np | Used for claimed discovery of element 43 |
| Dubna | Russium | Rs | Used for claimed discovery of element 43 |
Plausible names
Many speculative names appear in popular literature. The table below lists these names in the case where they obey IUPAC rules and are plausible with regard to the claimant laboratories. Rumoured suggestions (*) linked to the claimant laboratories are also included.
| Group Link | Suggested Name | Suggested Synbol | Derivation | Comments |
|---|---|---|---|---|
| RIKEN | Nihonium * | Nh | Nihon - Japanese for Japan | more appropriate, a really local name |
| Dubna | Becquerelium * | Bq | Henri Becquerel - discoverer of radioactivity | Dubna team suggested the name for element 110 |
Electronic structure
Ununtrium is element 113 in the Periodic Table. The two forms of the projected electronic structure are:
Bohr model: 2, 8, 18, 32, 32, 18, 3
Quantum mechanical model: 1s22s22p63s23p64s23d10 4p65s24d105p66s24f145d10 6p67s25f146d107p1
Extrapolated chemical properties of eka-thallium
Oxidation states
Element 113 is projected to be the first member of the 7p series of non-metals and the heaviest member of group 13 (IIIA) in the Periodic Table, below thallium. Each of the members of this group show the group oxidation state of +III. However, thallium has a tendency to form only a stable +I state du to the "inert pair effect", explained by the relativistic stabilisation of the 7s-orbitals, resulting in a higher ionisation potential and weaker tendency to participate in bonding.
Chemistry
Element 113 should portray eka-thallium chemical properties and should therefore from a monoxide, UutO, and monohalides, UutF, UutCl, UUtBr and UutI. If the +III state is accessible, it is likely that it is only possible in the oxide, Uut2O3, and fluoride, UutF3.
History of synthesis of isotopes by cold fusion
209Bi(70Zn,xn)279-x113 (x=1)
The synthesis of element 113 was first attempted in 1998 by the team at GSI using the above cold fusion reaction. In two separate runs, they were unable to detect any atoms and calculated a cross section limit of 900 fb. They repeated the experiment in 2003 and lowered the limit further to 400 fb. In late 2003, the emerging team at RIKEN using their efficient apparatus GARIS attempted the reaction and reached a limit of 140 fb. In December 2003-August 2004, they resulted to 'brute force' and performed an eight-month-long irradiation in which they increased the sensitivity to 51 fb. They were able to detect a single atom of 278113. They repeated the reaction in several runs in 2005 and were able to synthesise a second atom. They calculated a record-low 31 fb for the cross section for the 2 atoms.
History of synthesis of isotopes by hot fusion
237Np(48Ca,xn)285-x113 (x=3)
In June 2006, the Dubna-Livermore team synthesised ununtrium directly in the "warm" fusion reaction between neptunium-237 and calcium-48 nuclei. Two atoms of 282Uut were detected with a cross section of 900 fb.
Chronology of isotope discovery
| Isotope | Year discovered | discovery reaction |
|---|---|---|
| 278Uut | 2004 | 209Bi(70Zn,n) |
| 279Uut | unknown | |
| 280Uut | unknown | |
| 281Uut | unknown | |
| 282Uut | 2006 | 237Np(48Ca,3n) |
| 283Uut | 2003 | 243Am(48Ca,4n) |
| 284Uut | 2003 | 243Am(48Ca,3n) |
Yields of isotopes
Cold fusion
The table below provides cross-sections and excitation energies for cold fusion reactions producing ununtrium isotopes directly. Data in bold represents maxima derived from excitation function measurements. + represents an observed exit channel.
| Projectile | Target | CN | 1n | 2n | 3n |
|---|---|---|---|---|---|
| 70Zn | 209Bi | 279Uut | 31 fb |
Hot Fusion
The table below provides cross-sections and excitation energies for hot fusion reactions producing ununtrium isotopes directly. Data in bold represents maxima derived from excitation function measurements. + represents an observed exit channel.
| Projectile | Target | CN | 3n | 4n | 5n |
|---|---|---|---|---|---|
| 48Ca | 237Np | 285Uut | 0.9 pb , 39.1 MeV |